The European Union (EU) has admitted it is urgently developing a cutting-edge “fast vax” in anticipation of “pandemics on the horizon” including the mysterious “Disease X” which has been included on a list of “analyzed pathogens.”
At the forefront of this initiative is the innovative German-EU “Vaccelerate” program, actively developing a vaccine for the next pandemic-causing pathogen – before the pathogen (Disease X) has even been announced to the public.
Scientists affiliated with the Vaccelerate program unveiled their plan for a rapid vaccine in a paper titled “Predicting the Next Pandemic.” The paper delves into a comprehensive survey conducted among the EU scientific community, seeking insights into the pathogen most likely to trigger the “next pandemic” or serve as the next “potential pandemic generator.”
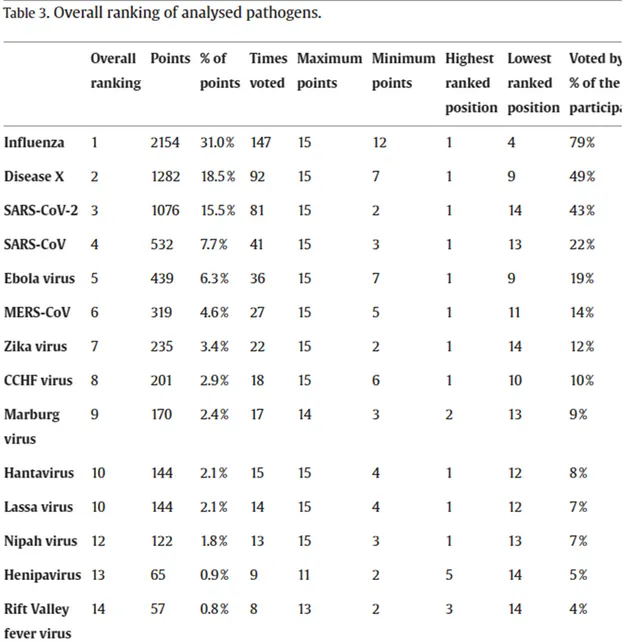
The survey responses, a collective ranking of various candidates, were meticulously tallied using a points system. Notably, influenza emerged as the top choice, endorsed by nearly 80% of respondents and securing almost one-third of the total points.
Taking second place was the ominous “Disease X,” an unknown entity in the realm of potential threats, while the well-known Coronaviruses, SARS-CoV-2 and SAR-CoV-1, claimed the third and fourth positions respectively. The detailed results are provided below.
The roots of the Vaccelerate program can be traced back to the European Vaccine Trial Accelerator Platform (EUVAP), initiated with a generous €600,000 grant from the German Government at the University of Cologne in 2020. This initiative gained momentum during the initial rush to develop a COVID-19 vaccine, as documented under “Vaccelerate.”
By early 2021, Vaccelerate became a major EU project, evolving into a pan-European network with substantial support from the European Commission. According to its EU funding page, the program has received an impressive total of €26.5 million in EU funding.
Outlined in the project description on the funding page, Vaccelerate’s original mission, aligned with EUVAP’s objectives, was to establish a comprehensive EU-wide network of clinical trial sites and a registry of willing participants. The goal was to expedite the development of COVID-19 vaccine candidates. Emphasizing the urgent need for a solution during the ongoing pandemic, the description highlights:
“Vaccine-induced immunity is the only promising solution. There is continued need for phase two and three vaccine trials to reach long-term, large-scale immunity of the entire European population.”
As the program’s overarching objective, Vaccelerate positions itself as the pan-European backbone propelling phase two and three COVID-19 vaccine trials forward. Remarkably, by the time Vaccelerate took center stage in 2021, the swift rollout of COVID-19 vaccines had already set a record for efficiency.
However, according to the EU, future vaccines must be rolled out even fast, and with less regulatory oversight, than the disastrous COVID-19 mRNA vaccines which are set to wreak havoc on public health around the world for decades to come.

















